 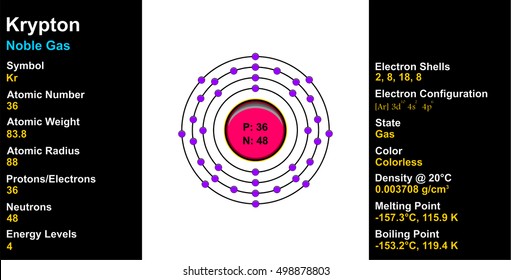
To find the number of neutrons in an atom, simply subtract the number of protons from the mass number. And the number of particles present in the nucleus is called the mass number (also called the atomic mass). 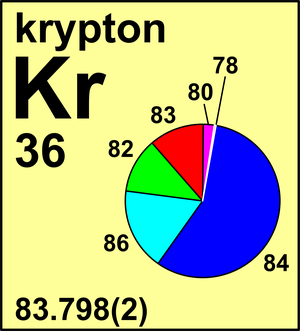
Which element has 51 neutrons? Antimony How do you find neutrons? Note that the atomic nucleus is made up of protons and neutrons. The three atomic particles in an atom are positively charged protons, negatively charged electrons, and uncharged neutrons. Are protons and electrons the same thing? In fact, the number of protons and electrons in an atom is the same only if the atom is neutral. The number of neutrons is equal to the difference between the ordinal number (M) and the ordinal number (Z). The mass number of the atom (M) is equal to the sum of the number of protons and neutrons in the nucleus. How do you calculate the electrons in an atom? The number of electrons in a neutral atom equals the number of protons. This becomes relevant in nuclear chemistry, where one sometimes has to weigh the central equations against mass numbers (numbers of nucleons). How many protons, neutrons and electrons does Krypton have with you? 36What does number 84 mean in krypton 84?In the case of Krypton84, this means you have 84 nucleons, 36 of which are protons and the rest 48 are neutrons. And how many protons, neutrons and electrons are there in a single krypton atom - 84? Krypton nameĪnd how many neutrons does Krypton 82 have? The mass number of the given atom is 82. This means that the nucleus of a Krypton84 isotope contains a total of 84 protons and neutrons. ISBN 978-0-8493-0485-9.How Many Neutrons Does Krypton Have How many protons and neutrons are there in an atom with a mass of 84 krypton? The mass number A for a certain isotope indicates how many protons Z, given by its atomic number, and how many neutrons it contains in the cell nucleus. CRC Handbook of Chemistry and Physics (85th ed.).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
